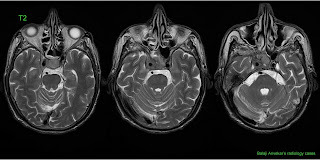
A 34 y o male with headache, nausea and vomiting.
Here is his MRI Brain, Axial T2, T1, FLAIR, Diffusion, Post contrast T1w images with MR Spectroscopy.
This MRI study of Brain shows:
Multiple intra axial round to ovoid unilocular cystic signal intensity focal lesions in supra tentorium as well as posterior fossa.
Fluid content of cyst is not very clear as it is hyperintense on FLAIR due to incomplete signal suppression of fluid, debris is seen in dependent portions.
The lesion in posterior fossa show no perilesional odema, lesions in parietal regions show mild odema underneath the lesion (may indicate an associated inflammation or a superadded infection).
None show restricted diffusion on DW images.
No significant mass effect.
Thin uniform thickness wall, non enhancing on post contrast T1w images.
On single voxel MR Spectroscopy at short TE of 35ms from right to left;
At 1.3 ppm - sharp doublets of lactate.
At 2.01ppm - no peak of NAA.
At 3.03ppm - no peak of Creatinine.
At 3.2ppm - no peak of Choline.
Imaging diagnosis : Hydatid cysts.
Follow up not available.
CEREBRAL HYDATID
Echinococcus (hydatid disease) affecting the CNS, is caused by E granulosis most commonly and E multilocularis.
Dogs or other carnivores are definitive hosts.
Sheep or cattle are intermediate hosts.
Humans are secondarily infected by ingestion of food or water contaminated with parasite eggs.
Parasite from GI tract to portal system, lymphatics.
Infection usually occurs in liver and lungs.
Typical imaging findings are large uni or multilocular cyst, isodense to CSF on CT and MRI. No edema and enhancement is typical. Fine peripheral enhancement may be seen on MRI. Calcification rare.
DDs
Neoplasm
Primary or metastatic (primary often known)
Thick, irregular margin enhancement typical.
Abscess
T2 hypointense rim and restricted diffusion is typical.
Perilesional odema.
Ring-enhancement.
Porencephalic cyst
Encephalomalacia +/- surrounding gliosis
Typically communicates with ventricle
Clinical presentation
Occur at all ages but commonly affect children and young adults.
Signs and symptoms depend up on location of the lesion.
Lesion develops slowly over many years. May get complicated by rupture, hemorrhage, secondary infection
Treatment
Variable, ranges from oral therapy to lesion resection.
Here is his MRI Brain, Axial T2, T1, FLAIR, Diffusion, Post contrast T1w images with MR Spectroscopy.
Multiple intra axial round to ovoid unilocular cystic signal intensity focal lesions in supra tentorium as well as posterior fossa.
Fluid content of cyst is not very clear as it is hyperintense on FLAIR due to incomplete signal suppression of fluid, debris is seen in dependent portions.
The lesion in posterior fossa show no perilesional odema, lesions in parietal regions show mild odema underneath the lesion (may indicate an associated inflammation or a superadded infection).
None show restricted diffusion on DW images.
No significant mass effect.
Thin uniform thickness wall, non enhancing on post contrast T1w images.
On single voxel MR Spectroscopy at short TE of 35ms from right to left;
At 1.3 ppm - sharp doublets of lactate.
At 2.01ppm - no peak of NAA.
At 3.03ppm - no peak of Creatinine.
At 3.2ppm - no peak of Choline.
Imaging diagnosis : Hydatid cysts.
Follow up not available.
CEREBRAL HYDATID
Echinococcus (hydatid disease) affecting the CNS, is caused by E granulosis most commonly and E multilocularis.
Dogs or other carnivores are definitive hosts.
Sheep or cattle are intermediate hosts.
Humans are secondarily infected by ingestion of food or water contaminated with parasite eggs.
Parasite from GI tract to portal system, lymphatics.
Infection usually occurs in liver and lungs.
Typical imaging findings are large uni or multilocular cyst, isodense to CSF on CT and MRI. No edema and enhancement is typical. Fine peripheral enhancement may be seen on MRI. Calcification rare.
DDs
Neoplasm
Primary or metastatic (primary often known)
Thick, irregular margin enhancement typical.
Abscess
T2 hypointense rim and restricted diffusion is typical.
Perilesional odema.
Ring-enhancement.
Porencephalic cyst
Encephalomalacia +/- surrounding gliosis
Typically communicates with ventricle
Clinical presentation
Occur at all ages but commonly affect children and young adults.
Signs and symptoms depend up on location of the lesion.
Lesion develops slowly over many years. May get complicated by rupture, hemorrhage, secondary infection
Treatment
Variable, ranges from oral therapy to lesion resection.