|
| MRI ANKLE |
Dr Balaji Anvekar FRCR
Neuro and MSK Consultant Radiologist
Wednesday 9 October 2024
Lateral hindfoot impingement MRI Ankle
Monday 17 June 2024
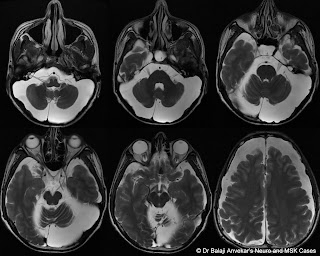
Dural ectasia spine MRI
Sagittal MR images of spine showing multiple contiguous widening of CSF space posterior to the cord in thoracolumbar region, associated posterior scalloping of the vertebral bodies, widening of caliber of the bony spinal canal, hypo plastic posterior elements.
Axial T2 images, STIR coronal images showing multiple lateral meningoceles in thoracolumbar region with enlarged neural foramen.
Spinal cord displaced anteriorly, flattened, compressed against anterior confines of anterior bony spinal canal.
Imaging diagnosis: dural ectasia.
Dural ectasia is widening of the dural sac, associated with herniation of nerve root sleeves out of foramina. Scalloping of the posterior vertebral body, thinning of cortex of pedicles and laminae, widening of neural foramina expected to prolonged pressure effect from the dural sac containing CSF.
Dural ectasia is very well-known with Marfan syndrome however can also be associated with other inherited connective tissue disorders, including Ehlers-Danlos syndrome and Loeys-Dietz syndrome, type 1 neurofibromatosis, ankylosing spondylitis, Lehman syndrome.
Dural ectasia can also be associated with trauma, scoliosis.
May be asymptomatic, may present with back pain, headaches, radicular pain, leg weakness or urinary incontinence.
Friday 19 April 2024
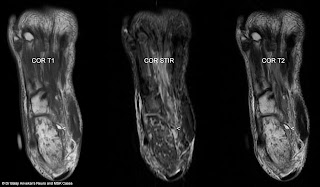
Little finger FDP tendon, A2 pulley injury
MRI LITTLE FINGER
 |
| SAGITTAL T2 5TH FINGER |
 |
| SAGITTAL T2 5TH FINGER |
 |
| AXIAL T2 A2 PULLEY AT MID PORTION OF PROXIMAL PHALYNX 5TH FINGER |
This MRI study of litter finger shows:
Full-thickness flexor digitorum profundus, FDP tendon tear, Zone II injury.
There is associated retraction of the proximal end, spring coiled, the torn end of proximal stump is at the level of metacarpo phalangeal joint, with a gap of 36 mm. Length of the distal stump measuring approximately 20 mm from its insertion on to the base of distal phalanx. Quality of both torn ends of the tendon is good, sharp without abnormal fraying or degeneration.
Associated A2 pulley injury at the level of midportion of proximal phalanx with approximately 4 mm bowstringing of FDS.
A3 pulley intact.
No associated bony avulsion.
No associated joint subluxation.
No associated volar plate injury.
No associated collateral ligaments tear.
Sunday 9 April 2023
Graefe Usher syndrome MRI
MRI brain shows:
Imaging diagnosis: Graefe-Usher syndrome.
Usher syndrome is characterized by partial or total hearing loss, vision loss that worsens over time. The hearing loss is sensorineural, caused by abnormalities of the inner ear.
A rare, congenital, autosomal recessive disorder characterized by retinitis pigmentosa and sensorineural hearing loss, first described by Von Graefe in 1858.
Saturday 25 March 2023
Mazabraud's syndrome
MRI with Xray correlation shows:
1. Mixed signal intensity lobulated lesion involving metadiaphysis of left proximal femur, part of adjacent epiphysis. Lesion is slightly expansile with groundglass matrix in the region of metaphysis on x-ray. No periosteal reaction on x-ray as well as MRI. No obvious pathological fracture. No abnormal adjacent bone marrow oedema on STIR.
Imaging wise possible diagnosis: Fibrous dysplasia.
2. Multiple T2 hyperintense lobulated space-occupying lesions involving muscles of left adductor compartment. The largest lesion measuring approximately 76 mm x 40 mm at a distance of 20 cm from greater trochanter on medial aspect of femur at 7 o’clock position on axial section.
Imaging wise possible diagnosis: intramuscular myxomas.
Intramuscular myxomas + left femoral fibrous dysplasia = Mazabraud's syndrome.
Saturday 24 December 2022
AVN collapse prediction by Modified Kerboul method
The arc of the femoral surface involved by necrosis measured by angles on midcoronal (A) and midsagittal image (B) and then modified Kerboul angle (A+B) calculated by the sum of the two angles for both the joints.
Right side the angle is (129+169) =298, grade 3 lesion.
GRADING
On the basis of combined angle, hips are classified into four categories:
Grade 1 (<200 degrees),
Grade 2 (200 degrees to 249 degrees),
Grade 3 (250 degrees to 299 degrees), and
Grade 4 (>/=300 degrees).
BACKGROUND
The hypothesis is that the combined necrotic angle measurement from magnetic resonance imaging scans predicts the subsequent risk of collapse in hips with femoral head necrosis.
With use of the modified method of Kerboul et al., Angle calculated by sum of the arc of the femoral surface involved by necrosis on a midcoronal as well as a midsagittal magnetic resonance image calculated on MRI, rather than on an anteroposterior and a lateral radiograph is far more accurate than on X-ray.
Friday 4 November 2022
Physeal bony bar MRI
Tuesday 12 July 2022
Extramedullary focal fat - fluid level, a specific sign of osteomyelitis
A 14 yo male with pain in calcaneum since 1months.
MRI foot for calcaneum with CT correlation shows:
Heterogeneous signal abnormality diffusely involving calcaneum with multiple low signal intensity foci diffusely scattered in calcaneum on T1-weighted images which are hyperintense on STIR. Rest of the intervening calcaneum medulla shows faint high signal on STIR.
No obvious density abnormality on CT. No obvious sclerotic or lytic lesion. No obvious cortical destruction or sclerosis.
There is a focal lentiform shaped parosteal collection measuring approximately 26 mm in height and 6 mm in thickness medially at 2 o’clock position and 4 mm in thickness laterally on plantar aspect at 7 o’clock position on axial section.
There is fat – fluid level within this collection, focal fat in the supernatant portion of this collection which is hyperintense on T1-weighted images with complete signal suppression on STIR, this portion follows classical fat density on CT.
There is an associated diffuse oedema involving muscles of plantar aspect of foot especially quadratus plantae muscle, medial as well as lateral teno synovitis.
This finding suggestive of extramedullary focal fat - fluid level which is a pathognomonic sign of acute to subacute osteomyelitis.
Findings were discussed with the referring physician before finalizing the report, who added that there is elevation of inflammatory markers in lab reports and the suspicion of osteomyelitis clinically as well, with a feedback of significant improvement clinically after IV antibiotics.
Take home note is during MSK MRI interpretation, a bone marrow signal abnormality with an associated focal periosteal extra medullary fat – fluid level, osteomyelitis should be in the list of your differential diagnosis.
References:
1. Extra-osseous fat fluid level: a specific sign for osteomyelitis. Kumar J, Bandhu S, Kumar A, Alam S. Skeletal Radiol. 2007 Jun;36 Suppl 1:S101-4. doi: 10.1007/s00256-006-0194-1. Epub 2006 Sep 19.
2. Intramedullary and extramedullary fat globules on magnetic resonance imaging as a diagnostic sign for osteomyelitis. Davies AM, Hughes DE, Grimer RJ. Eur Radiol. 2005 Oct;15(10):2194-9. doi: 10.1007/s00330-005-2771-4. Epub 2005 Apr 29.
Sunday 17 October 2021
Rice bodies in subdeltoid bursa MRI
This MRI shoulder joint shows fluid distended sub deltoid bursa with numerous typical rice bodies.
Elbow Neuropathic Arthropathy MRI
A middle aged male with unilateral left elbow pain, deformity, progressive swelling, restricted movement.
This MRI elbow joint shows:
Joint effusion, osteolysis involving proximal end of radius as well as ulna, articulating surface of capitulum as well as trochlea. Marked synovial thickening with frond like projections.